 
Topical TofacitinibPatient characteristics are summarized in Table 2. 2 The appeal of topical JAKi is potentially a reduced side effect profile through a different mode of entry. In this article we will cover the most prominent therapeutic candidates in the alopecia areata treatment industry in 2023. One patient stopped treatment due to new-onset multiple sclerosis. While the use of oral JAKi has shown potential as a successful treatment for AA, oral JAKi have been associated with side effects, such as infections, viral reactivation and rarely thromboembolic events. 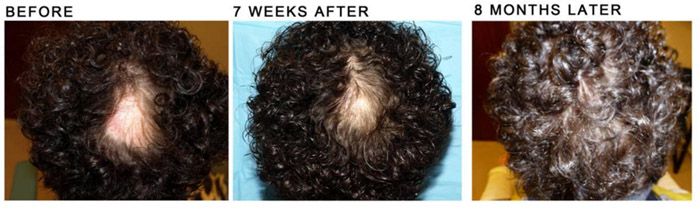
Other side effects included gastrointestinal symptoms and mild acne. One patient developed hyperlipidemia and weight gain while on 11 mg extended release twice daily, which improved with exercise and diet changes while remaining on treatment. Of the ten patients with documented follow-up SALT scores, five patients achieved complete disease resolution (SALT score <=5%), three of whom were treated with adjuvant ILK treatment (5-10 mg/ml) to recalcitrant patches.Five patients temporarily ceased treatment and were observed to have gradual patchy loss of hair regrowth. One patient was not included because she was lost to follow up. The mean SALT score improvement from baseline was calculated to be 61.18% (n=10, range, 0%-100%). Patients were treated with oral tofacitinib (range 5 mg once daily to 11 mg extended release twice daily) for a mean treatment duration of 14.4 months (range, 4.5-27 months).The mean time to first response was 1.36 months (range 4 days to 3 months). We identified 10 patients with alopecia areata universalis and one patient with alopecia areata totalis with mean duration of disease of 5.23 years (range, 3-11 years). Oral TofacitinibPatient characteristics are summarized in Table 1. The % SALT score change from baseline was calculated as (initial SALT score - best SALT score on treatment)/initial SALT score x 100%. The SALT score was calculated prior to initiation of treatment and throughout follow-up by a combination of in office physician evaluation and retrospective photographic evaluation. The review spanned Apto August 20, 2017.Treatment response was evaluated by patient reported time to initial response (defined as time to first documented sign of any hair regrowth) and the validated Severity of Alopecia Tool (SALT) score, with a higher score indicating more severe disease. This case series was approved by the University of California, Los Angeles Institutional Review Board. Notably, two of these patients were treated with oral tofacitinib either prior to or after topical tofacitinib, but not concurrently with topical tofacitinib (Table 2). Additionally, we identified four patients with severe AA who were treated with 2% tofacitinib cream twice daily. Adjuvant intralesional Kenalog (ILK) was administered at the physician’s discretion. Dosage was titrated based on response, tolerability, and insurance coverage. Oral tofacitinib treatment was started at 5 mg once daily, 5 mg twice daily, or 11 mg extended release once daily. Eleven patients diagnosed with AA universalis or totalis treated with oral tofacitinib were identified (Table 1). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
